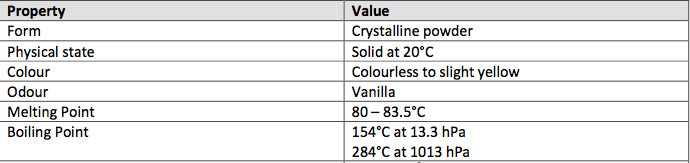
Physical Properties
Chemical Properties
Reactivity: Vanillin can react violently with Br2, HClO4, potassium-tert-butoxide.
Reactivity with Air: Slowly oxidizes on exposure to air.
Flammability: not highly flammable but still possible
Toxicity: Not toxic
Solubility: Slightly water soluble.
Reactivity with Air: Slowly oxidizes on exposure to air.
Flammability: not highly flammable but still possible
Toxicity: Not toxic
Solubility: Slightly water soluble.
Relationship between properties and structure
Melting and Boiling Point: Vanillin is a molecular compound and therefore there is not an electron transfer but it has shared electrons. The bond in ionic compounds is much stronger than in molecular compounds therefore vanilliin will have a lower melting and boiling point compared to ionic bonds because it is easier to break the electrons apart.
Reactivity: Vanillin has a highly reactive carbonyl group in its structure. When mixed with Br2, HClO4, and potassium-tert-butoxide the carbonyl group becomes satisfied and reacts violently.
Solubility: Because the O - C and the single O - H bonds are polar but the other bonds in the structure are not polar the water only attracts to the dipoles of the O - C and the O - H bond making it slightly soluble.
Reactivity: Vanillin has a highly reactive carbonyl group in its structure. When mixed with Br2, HClO4, and potassium-tert-butoxide the carbonyl group becomes satisfied and reacts violently.
Solubility: Because the O - C and the single O - H bonds are polar but the other bonds in the structure are not polar the water only attracts to the dipoles of the O - C and the O - H bond making it slightly soluble.